Quiz - Understanding Molecules and Bonds
Number of questions :
8
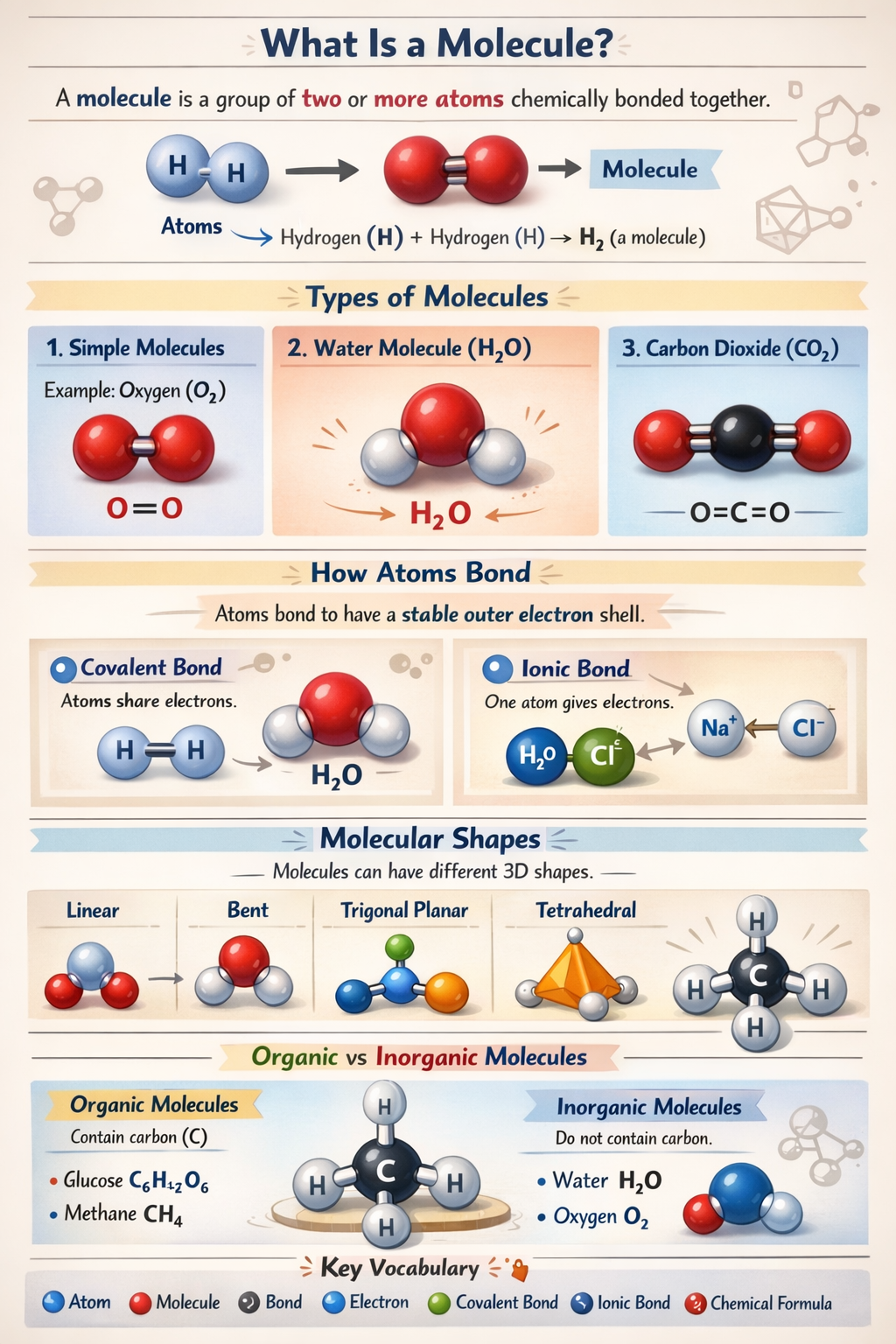
This infographic explains what a molecule is, types of molecules, how atoms bond, molecular shapes, and the difference between organic and inorganic molecules. It covers basic concepts such as covalent and ionic bonds, and provides examples like water, oxygen, and carbon dioxide.
Understanding molecules and their bonds is fundamental to chemistry and science.
- By learning about molecules, you gain insight into the basic building blocks of matter. This knowledge is crucial for fields like biology, physics, and environmental science. Understanding how atoms bond to form molecules helps explain the properties and behaviors of different substances.
- Covalent and ionic bonds are essential concepts that explain how atoms interact. Covalent bonds involve sharing electrons, while ionic bonds involve transferring electrons. These interactions determine the stability and reactivity of molecules.
- Molecular shapes influence how molecules interact with each other. For example, the bent shape of water molecules leads to its unique properties, such as surface tension and solvent abilities.
- Organic vs. inorganic molecules: Knowing the difference helps in understanding biological processes and the composition of various materials. Organic molecules contain carbon and are vital for life, while inorganic molecules are found in non-living systems.
- This quiz enhances your comprehension of these concepts, making you more informed about the natural world and better prepared for advanced studies in science. It fosters critical thinking and problem-solving skills, which are valuable in both academic and real-world scenarios.
You may be interested in these quizzes
Understanding Molecules and Bonds
Chemistry
Science Education
Molecular Structure
14/02/2026
This infographic explains what a molecule is, types of molecules, how atoms bond, molecular shapes, and the difference between organic and inorganic molecules. It covers basic concepts such as covalent and ionic bonds, and provides examples like water, oxygen, and carbon dioxide.
Number of questions : 8
Chemistry Exam: Key Concepts
Chemistry
Physics
Science Education
06/01/2026
This document is a chemistry exam covering topics such as atomic structure, chemical bonding, molecular geometry, and gas laws. It includes multiple-choice questions that test knowledge of periodic table elements, VSEPR theory, hybridization, and thermodynamic principles.
Number of questions : 8
Chemical Reactions Educational Poster

Chemistry
Science Education
Laboratory Safety
05/01/2026
This document is an educational poster about chemical reactions, covering topics such as reactants and products, signs and types of chemical reactions, conservation of mass, chemical equations, and safety rules in the lab.
Number of questions : 8
Periodic Table of Elements Overview
Chemistry
Science
Education
24/02/2026
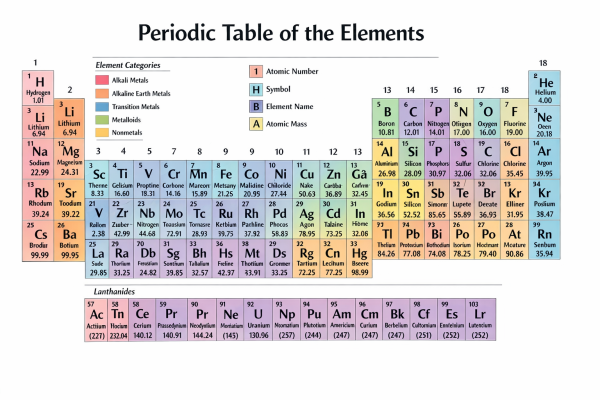
The periodic table organizes chemical elements by increasing atomic number, grouping them into categories such as alkali metals, transition metals, and nonmetals. It provides essential information like atomic mass and element symbols, serving as a fundamental tool in chemistry for understanding element properties and relationships.
Number of questions : 8
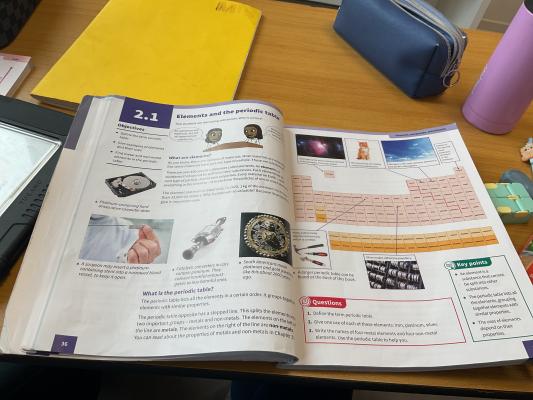
Elements and Periodic Table Overview
Chemistry
Education
Science
30/01/2026
This document is an educational page about the periodic table and elements. It explains the definition of elements, the importance of platinum, and the structure of the periodic table. It also includes examples of metals and non-metals and their uses in everyday life.
Number of questions : 8
Respiration Rate Measurement Apparatus

Biology
Chemistry
Scientific Methods
29/03/2026
This document is about measuring the respiration rate of small animals like woodlice using a specific apparatus. It explains the role of potassium hydroxide in absorbing carbon dioxide and the function of a syringe in the setup.
Number of questions : 8
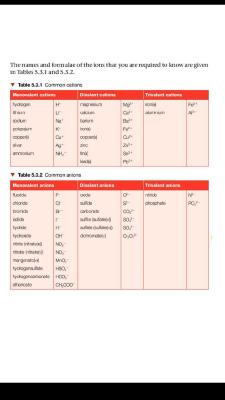
Common Cations and Anions
Chemistry
Ions
Chemical Formulas
08/03/2026
The document provides tables of common cations and anions, including their names and chemical formulas. It categorizes ions into monovalent, divalent, and trivalent types, essential for understanding chemical reactions and compounds.
Number of questions : 8